VISCOJECT VET
A device for injecting an intraocular lens (IOL) into an eye.
Dedicated guiding rails define the correct loading position of the IOL and the loading cartridge ledges lock the IOL in place, securing the IOL during the closure of the cartridge.
Assistance with separate loading block
The enclosed loading block secures cartridge positioning during IOL loading. The cartridge can be easily inserted into the injector after loading while the IOL stays secured.
- Description
Description
DESCRIPTION
The VISCOJECT™ VET injection system permits the implantation of foldable one-piece veterinary intraocular lenses through incisions of about 1.8 to 2.8mm, depending on the
VISCOJECT™ VET model and operating technique used, thanks to the new VISCOJECT™ VET hydraulics system. With the VISCOJECT™ VET injection system the majority of all foldable, one-piece acrylic lenses with an optical diameter of 6.5mm can be injected. The VISCOJECT™ VET injection system consists of three parts:
1. The sterile VISCOJECT™ VET single-use injector with silicone tip
2. The loading unit
3. The sterile VISCOJECT™ VET single-use cartridge
APPLICATION
Folding and injecting of one-piece foldable veterinary lenses into the
capsular bag or sulcus after extra-capsular cataract extraction.
INSTRUCTIONS FOR USE
1. Open the blister in the sterile area and remove the sterile VISCOJECT™ VET injector set.
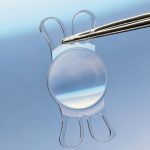
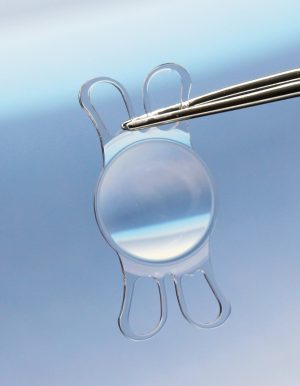
2. VISCOJECT™ VET cartridges are equipped with guiding rails. This allows rapid and precise loading of the lenses. Hold the loading unit with the VISCOJECT™ VET cartridge in such a manner that the rear cartridge flank can be guided with the index finger of the left hand. Position the rear cartridge flank so that the distance between the guiding rails corresponds approximately to the size of the lens optic.
3. Push the lens onto the guide rails from the rear and position it in the middle of the loading chamber.
4. Apply a small amount of viscoelastic material directly to the opening of the cartridge tunnel, as well as a sufficient quantity into the loading chamber below the lens. This ensures that the “VISCOJECT™ effect“ comes to bear.
5. Close the cartridge on the loading unit (the lens automatically folds into the right direction) until the click-lock mechanism engages. Carefully remove the cartridge from the loading block. The lens should be injected within one to two minutes after loading. Viscoelastic materials may lose their lubricity if allowed to stand too long while exposed to air.
6. Guide the cartridge into the opening at the front end of the injector and push it to the very front position.
7. Carefully push the plunger forwards and ensure that the silicone tip correctly enters the loading chamber. If the silicone tip cannot be introduced into the loading chamber, retract the plunger to the starting position and align the silicone tip straight using sterile tweezers. Continue to push the cushion until the inner spring begins to press together. IMPORTANT: Pull the plunger back a few millimetres and then push it forward again. This step ensures that the lens is correctly grasped.
8. Press the lens to the front part of the cartridge point outside of the eye. This prevents pressure generated by the
VISCOJECT™ VET system and an increased quantity of viscoelastic solution from being transferred to the eye.
9. Guide the point of the cartridge through the incision and push it across the iris to the near edge of the pupil.
10. Slowly press the plunger forward. Depending on the lens model, support the correct ejection of the lens by slightly turning the injector. Slowly inject the lens into the eye and simultaneously withdraw the instrument from the eye. To avoid swelling of the silicone tip during ejection from the cartridge, only push the plunger until the lens has emerged completely, even if the plunger is not yet at the limit.
11. Position the lens carefully and if necessary rotate it with the aid of the appropriate positioning hook.
12. Remove the viscoelastic material from the eye and the lens with the standard irrigation and aspiration techniques.
PLEASE ENSURE:
Do not re-sterilize / prepare VISCOJECT™ VET injections system. Single use only. Reuse and/or resterilization may compromise device performance, which could cause serious harm to the patient’s health and safety. Store at room temperature.